

Leading the pharmaceutical industry to become a superior provider of pharmaceutical products and services.
Adhere to R&D innovation and international expansion.

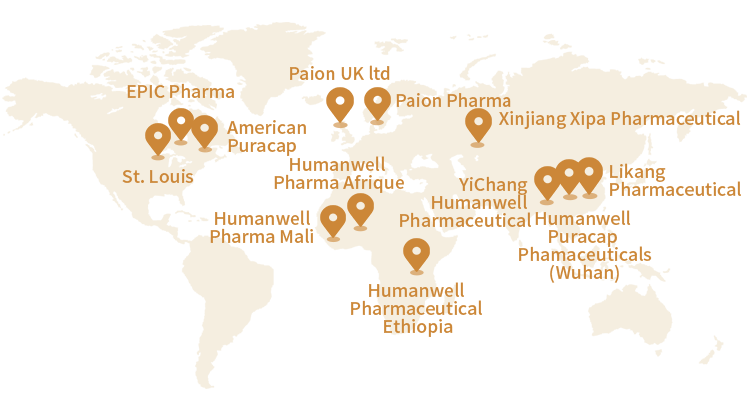
EPIC Pharma
St. Louis Research and Development Center
PuraCap Pharmaceutical
Humanwell Pharma Mali
Humanwell Pharma Afrique
Humanwell Pharmaceutical Ethiopia
YiChang Humanwell Pharmaceutical
Humanwell Puracap Phamaceuticals (Wuhan)
Likang Pharmaceutical
Paion Pharma
Paion UK
Xinjiang Xipa Pharmaceutical
尊龙凯时,尊龙凯时医药集团股份公司官网,尊龙凯时医药集团股份公司,尊龙凯时甾体激素,尊龙凯时麻醉药品医药2025年第三季度报告
Download尊龙凯时,尊龙凯时医药集团股份公司官网,尊龙凯时医药集团股份公司,尊龙凯时甾体激素,尊龙凯时麻醉药品医药2025年半年度报告
Download尊龙凯时,尊龙凯时医药集团股份公司官网,尊龙凯时医药集团股份公司,尊龙凯时甾体激素,尊龙凯时麻醉药品医药2025年第一季度报告
Download尊龙凯时,尊龙凯时医药集团股份公司官网,尊龙凯时医药集团股份公司,尊龙凯时甾体激素,尊龙凯时麻醉药品医药2024年年度报告
Download